Pharma and Biotech
Perspectives and best practices from the world of healthcare and life sciences.
Clarifying clinical trial results
For brands, communicating clinical trial results is usually paramount—the clinical efficacy data, balanced by the side effects observed, is the foundation of the product’s value proposition for patients. But clinical trial data is typically complex, and can be difficult for patients to understand.
Redesigning Important Safety Information (ISI) for advanced therapies
Important Safety Information appears throughout branded patient marketing materials. It captures key information that patients with advanced and complex diseases want to know, and supports informed consent for important treatment decisions. Countless hours of co-creation time with patients, caregivers, HCPs, and subject matter experts has helped us think differently about how we can communicate ISI to patients.
Thinking holistically about the patient experience
Patients with rare and complex diseases need brand marketing materials that offer concrete support. That’s why our approach is built on clear and effective communication, holistically integrating UX, visuals, clinical trial data, and important safety information.
Human-centered systems thinking: lessons from the Sepsis Watch rollout
Back to Ideas Some time ago, we came across an insightful article in the MIT Technology Review discussing the rollout of Sepsis Watch, a...
Form design 101
The experience of using and designing a form – an integral step to providing care that shapes the overall patient experience
On journey mapping
Moving beyond customer-centricity theatre. An in-depth breakdown of journey maps and how to use them correctly.
The case for early investment in rare disease state education
An examination of reasons why disease state education for rare diseases helps patients better navigate their journey and complex decisions.
Why building awareness for rare disease clinical trials matters
A framework that can help us better measure patient experience, and allow organizations to continually learn, grow, and implement changes both small and large to improve patients’ experiences.
5 tips for safely using LLMs to get medical advice
Examining issues with using LLMs to gather health information, and tips for having useful and less risky conversation with LLMs.
Improving patient experience: a multidimensional measurement framework
A framework that can help us better measure patient experience, and allow organizations to continually learn, grow, and implement changes both small and large to improve patients’ experiences.
The participation problem: addressing socioeconomic barriers in clinical trials
Genetics and socioeconomic factors can influence how a drug performs, and yet, a persistent gap remains between the people who need new treatments and those represented in the clinical trial data.
Women’s representation in clinical research
An examination of the gap in understanding women’s health by looking back at women’s representation in clinical research over time.
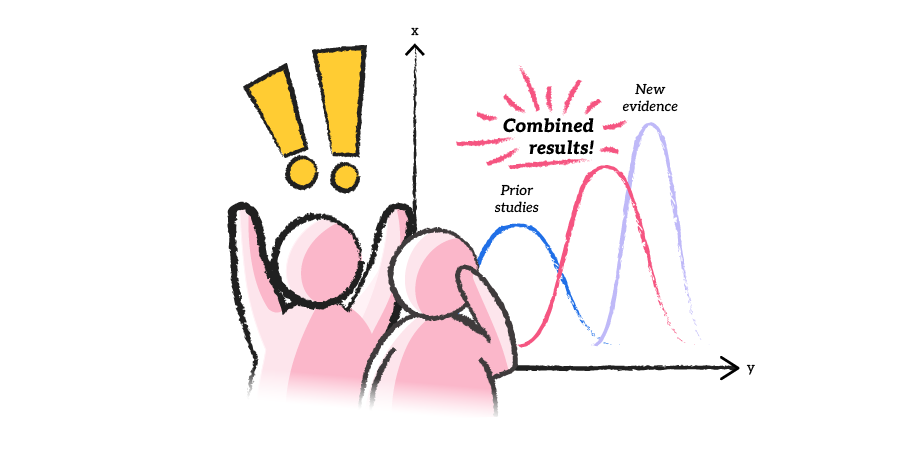
A new approach to clinical trials
A look a how Bayesian analytics could open the doors to cheaper and faster clinical trials
Hybrid co-creation in pharma and biotech
Bringing internal participants together for a hybrid co-creation creates shared intentions and speed to market.
Informed consent in healthcare: adding to FDA guidance
We share best practices in patient communication that may help comply with FDA guidance on informed consent, as well as practical tips.
HCP communication best practices
HCP communications from the pharma industry need to be more focused, trustworthy, and multipurpose, to deliver value in the clinical setting.
Patient-centred communications in pharma and biotech
The Universal Patient Language (UPL) is an example of how a pharma company can create effective patient communications.
Treatment decision-making: a Framework
Commercial pharmaceutical teams should examine an array of factors that shape treatment decision-making to influence prescribing behaviour.
Once a month, we send out a newsletter of work-related ideas. Get our next edition delivered to your inbox.