On January 12, the FDA shared draft guidance that may fundamentally speed up how new medicines reach patients through clinical trial analysis. Based on our reading of Pratik Pawar’s Vox article, the guidance may also affect how researchers design future clinical trials and may have a potentially positive impact on patients.
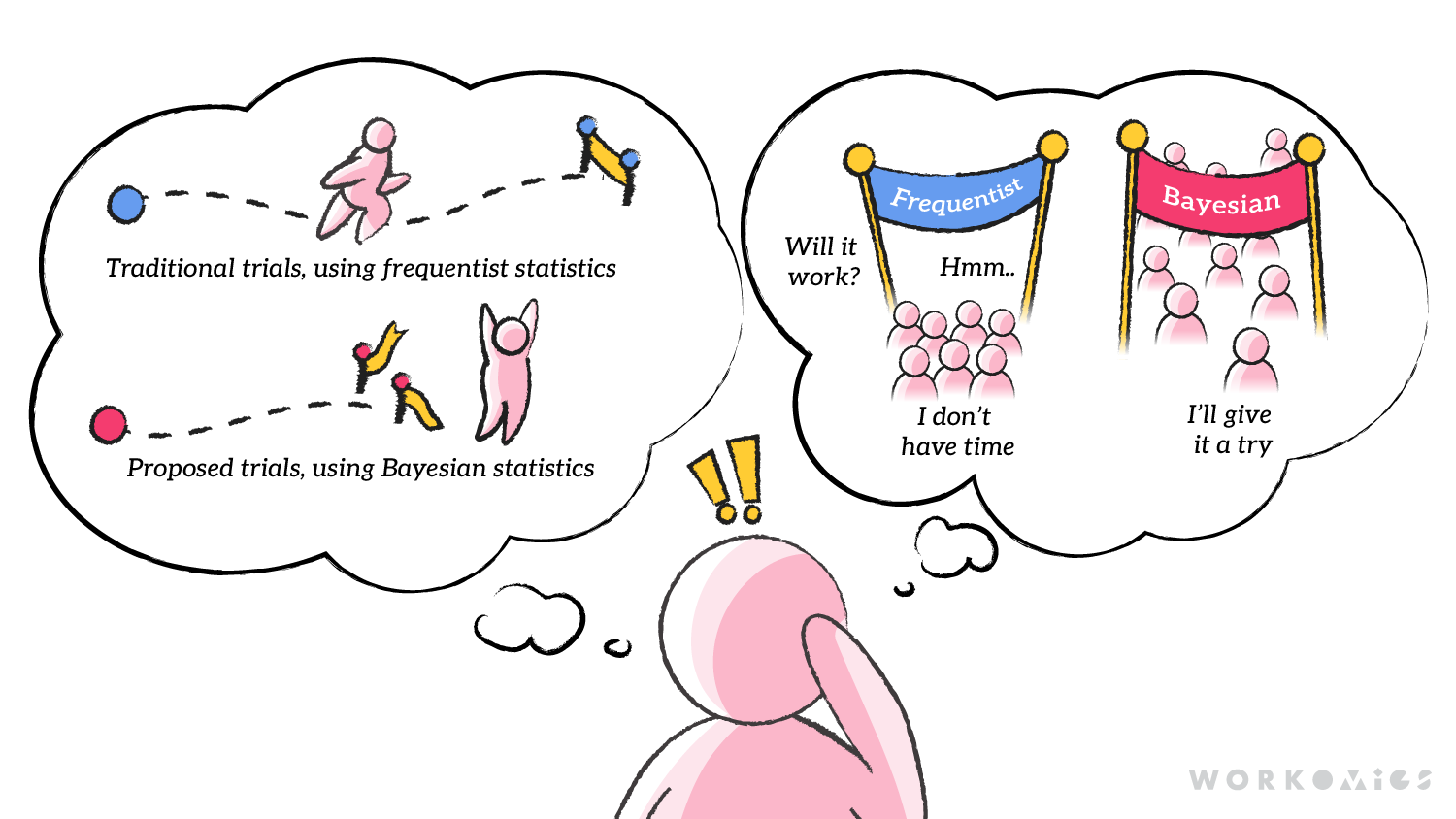
The FDA’s draft guidance document provides guidelines for industry on using Bayesian methodologies in clinical trial data when submitting INDs, NDAs, and BLAs. The rationale for these changes by the FDA is to reduce research costs and timelines, enabling patient access to effective treatments sooner. According to Pawar, the difference between the current and newly suggested analysis methods is that with frequentist statistics, clinical trials had to ensure that each trial’s data stood up on its own, answering the question “How likely would we see strong results about this drug, if the drug doesn’t work?” Bayesian methods, on the other hand, ask the question “Based on everything that is already known, how likely is it that the drug works?”
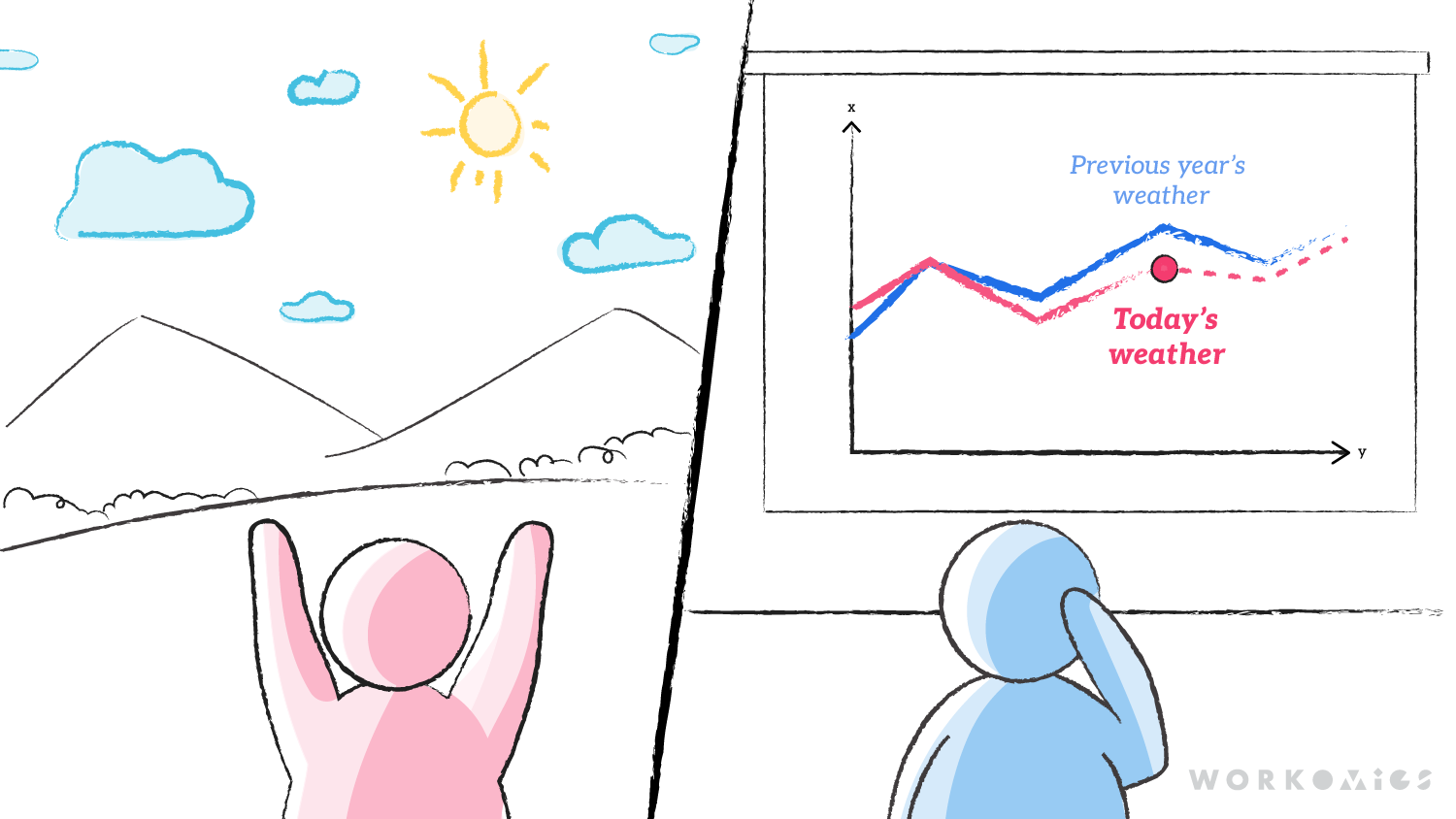
We like to think through things in analogies, and found comparing the new guidance on clinical trial analysis to weather prediction useful: if frequentist statistics are about predicting tomorrow’s weather using your own observations of today’s data (looking out the window, checking a thermometer), Bayesian analytics uses past data, or data from similar locations, to provide a more comprehensive view of what tomorrow’s weather might be (looking at average weather from past years, looking at what’s happening in towns at similar latitude).
There are, of course, potential risks with this approach, as past data could be cherry-picked to support desired results. The FDA’s guidance seeks to account for that by requiring a detailed listing of success criteria, assumptions, synthesis process, and modelling approaches in the trial protocol to prevent that. Pawar also notes that “with FDA facing leadership turnover and political uncertainty, companies may be even more cautious about trying something new.”
For a more in-depth look at what’s included in the guidance, you can read the draft guidance right here.
Our other ideas worth exploring
On journey mapping
Moving beyond customer-centricity theatre. An in-depth breakdown of journey maps and how to use them correctly.
The case for early investment in rare disease state education
An examination of reasons why disease state education for rare diseases helps patients better navigate their journey and complex decisions.
5 tips for safely using LLMs to get medical advice
Examining issues with using LLMs to gather health information, and tips for having useful and less risky conversation with LLMs.