Sponsors of late-stage clinical trials face a clear mandate from the FDA: they must assess safety and efficacy in populations that actually reflect the real world. Yet, despite these requirements, a persistent gap remains between the people who need new treatments and those represented in the data.
The numbers tell a sobering story. While the U.S. Census shows a population that is roughly 40% minority—including 19% Hispanic/Latino and 14% Black communities—clinical trial participation hasn’t kept pace. A 2022 analysis of 32,000 trial participants showed that only 8% were Black and 11% were Hispanic.*
This isn’t just a matter of “checking a box” for equity; it’s a scientific necessity. We know that genetics and socioeconomic factors can influence how a drug performs. If our study cohorts are narrow, our understanding of the product’s real-world impact remains incomplete. Furthermore, lower-income patients—who often face a higher burden of chronic disease—are frequently sidelined by time constraints or rigid trial designs, leaving researchers and trial sponsors without a full picture of the very people they aim to help.

Insights to Inform a More Inclusive Approach
A 2024 report, The Influence of Socioeconomic Status on Individual Attitudes and Experience with Clinical Trials, offers critical clues on how we can do better. The report highlights a few key trends:
The Education and Employment Gap
The study reports that Education, employment status, insurance coverage, and English language proficiency were important predictors of a person’s interest in participating in a clinical trial. Individuals with lower levels of attained education were less likely to have been asked to take part in a study, and were reported to be less interested in taking part in a clinical trial. Meanwhile, people who were unemployed also reported to be less interested in taking part in a clinical trial.
The Trust Barrier
Across all income and education levels, feeling like they would be “part of a medical experiment” was the top reason for patients not participating in a clinical trial. This was followed by concerns about medical procedures, cost, worries about one’s health, and lack of time. Concerns around being part of a medical experiment and medical procedures were most strongly associated with lower income respondents.
The Awareness Channel Shift
While email was the most common method via which folks were asked to participate in a clinical trial, the second-ranked method differed by race: white respondents cited hearing about a clinical trial from their healthcare provider, whereas Black, Asian, and Latino respondents cited social media. Relatedly, people who had a healthcare professional or former clinical trial participant in their personal network were more likely to have been asked to participate in a clinical trial.
How We Help our Clients Better Engage Underrepresented Communities
These findings echo what we see in our work every day: to build a more diverse and representative trial, you have to mind the details, establish a foundation of trust, and find ways to connect and engage with different communities. We think co-creation is an excellent starting point; it is at the heart of everything we do. This approach helps us create educational resources, including clinical trial materials that will better resonate with patients and their families, and be more likely to be used by clinicians with their patients at the point of care. In addition, a few specific strategies that we have applied to help close the gaps around underrepresented communities in clinical trials are listed below. A case study that showcases our approach in practice can also be seen here.
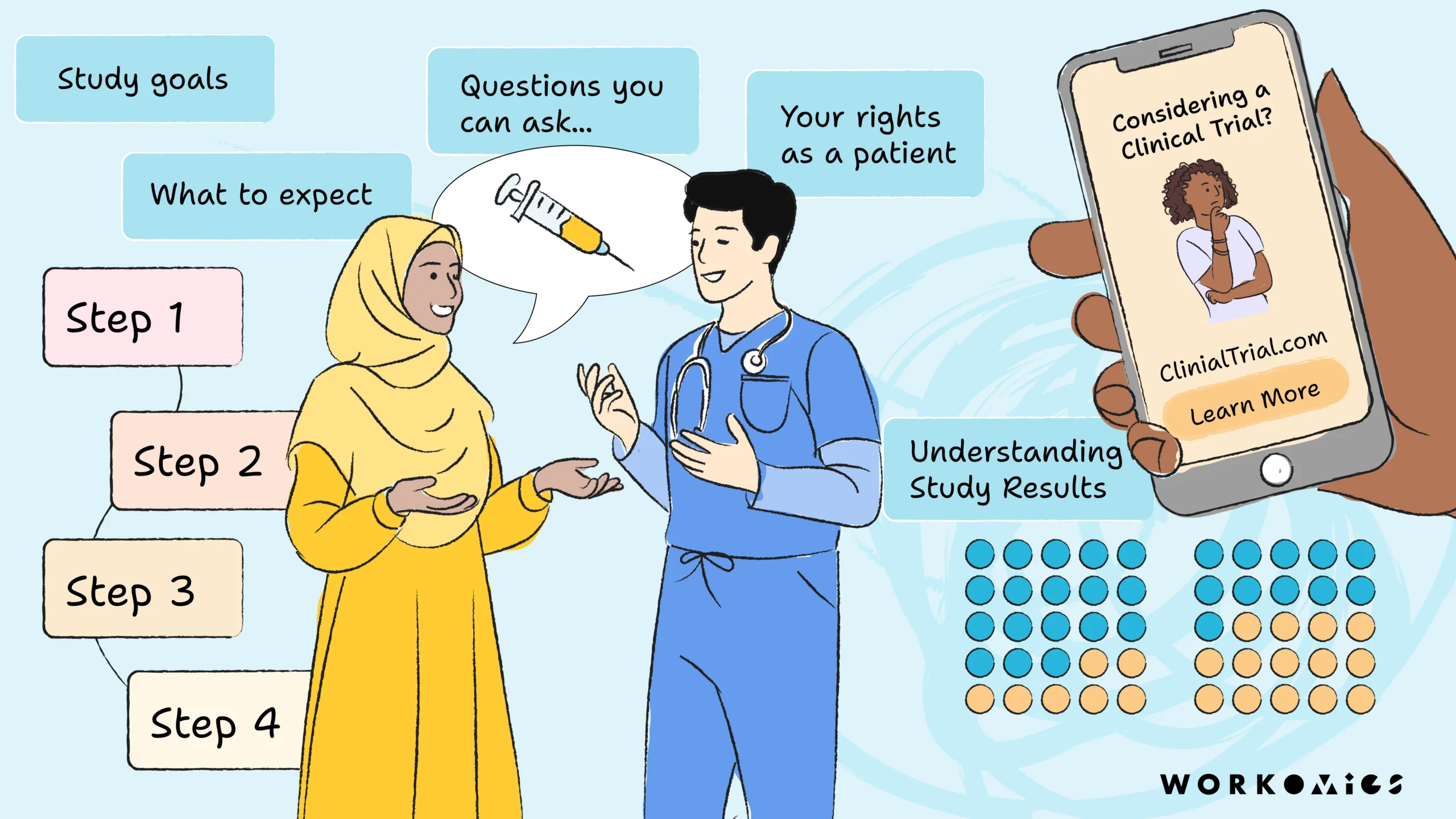
We prioritize representative imagery and plain language:
When we design clinical trial recruitment materials, we make them look and sound like the people they serve. This means using inclusive imagery of both patient and professional figures, and explaining complex medical terms and concepts that patients might encounter in the clinical setting.
We address the trust deficit through clear, transparent information
We demystify the clinical trial process by being transparent and clear around study goals, steps and methods, patient rights and protections. We directly acknowledge and answer the concerns that patients considering a trial might have, and suggest other questions for patients to take up with their doctors. Additionally, for people who use screen readers, we create accessible PDFs with proper reading order and ALT text. When feasible, we work with partners to create patient-facing resources in .
We meet people where they are, optimizing the right channels
Some communities rely more on social media than providers, and so we look for opportunities to reach patients there. We help our clients execute recruitment strategies with printed materials, websites and digital resources, as appropriate. In rare disease spaces, we’ve used low cost paid media strategies to help us reach hard-to-reach patients.
By shifting our focus from simple recruitment to intentional, inclusive engagement, we can move closer to a future where clinical research truly serves everyone. It’s a long-term commitment to better science—and ultimately, better patient outcomes.
*A source worth reading, “Inclusion and diversity in clinical trials: Actionable steps to drive lasting change,” can be found here.
Our other ideas worth exploring
On Journey Mapping
Moving beyond customer-centricity theatre. An in-depth breakdown of journey maps and how to use them correctly.
Why building awareness for rare disease clinical trials matters
A framework that can help us better measure patient experience, and allow organizations to continually learn, grow, and implement changes both small and large to improve patients’ experiences.
Improving Patient Experience: A multidimensional measurement framework
A framework that can help us better measure patient experience, and allow organizations to continually learn, grow, and implement changes both small and large to improve patients’ experiences.